 
Williams, Frangoul, and other experts in the field who expressed optimism about the future of gene therapy, despite relatively slow progress and new obstacles.Īll gene therapies are categorized as either in vivo or ex vivo. While these data suggest that the vector was not causative, “it is very important that this be investigated thoroughly, to better understand the cause of those complications that have happened years after therapy,†said Haydar Frangoul, MD, a pediatric hematologist at Tristar Medical Group in Nashville, Tennessee.ĪSH Clinical News spoke with Drs. The pharmaceutical company’s investigation has so far shown no link between cancer and the vector, but, as of mid-April, the trials have not resumed. In February 2021, trials of bluebird bio’s LentiGlobin gene therapy product for patients with sickle cell disease were paused after two patients developed serious illnesses, acute myeloid leukemia (AML) and myelodysplastic syndromes (MDS). However, there have been some recent setbacks for scientists testing gene therapies to treat hematologic malignancies and other blood disorders. €œThe progress that we’re seeing in the field is a testament to the investment in basic science research that preceded the clinical applications by many years,†said David Williams, MD, 2015 American Society of Hematology (ASH) President and a pediatric hematologist at Boston Children’s Hospital and Harvard Medical School whose research focuses on gene transfer methods. It took years, but researchers have now developed a variety of new gene therapy techniques, several of which have been brought to clinical trial. The progress being made by gene therapy researchers came to a screeching halt. You didn’t want to say, ‘I’m a gene therapist’ or ‘I’m working on gene therapy.’ It sounded terrible.†Even the term gene therapy became kind of a black label. 2 “That made the whole field of gene therapy go away, mostly, for at least a decade. €œWe were all very much aware of what happened there and what a tragedy that was,†commented Jennifer Doudna, PhD, who won the Nobel Prize in Chemistry in 2020 for her discovery of the CRISPR gene-editing tool. Within four days, he was declared brain dead. He developed a blood clotting disorder almost immediately and inflammation shut down several of his organs. 
Around the same time, Jesse Gelsinger, an 18-year-old with the rare metabolic disorder ornithine transcarbamylase deficiency syndrome, volunteered to be the 18th patient injected with an adenovirus that carried a normal ornithine transcarbamylase gene to treat his disease. It turned out that the viral vector that delivered the gene to their T cells also had activated an oncogene. The trial seemed successful, until five of the 20 patients developed leukemia. Patients with a similar disease (SCID-X1) received a comparable gene therapy in the late 1990s. Some early trial participants who came after her did not fare as well. Turning to a cutting-edge treatment, scientists were able to deliver to Ashanthi a healthy version of the gene that produces ADA using a viral vector. However, few patients achieve full immune reconstitution and improvements in immune function can wane in just a few years with enzyme replacement therapy. In addition to hematopoietic cell transplantation, standard treatment at that time consisted of lifelong replacement with synthetic ADA given via intramuscular injections once or twice weekly. 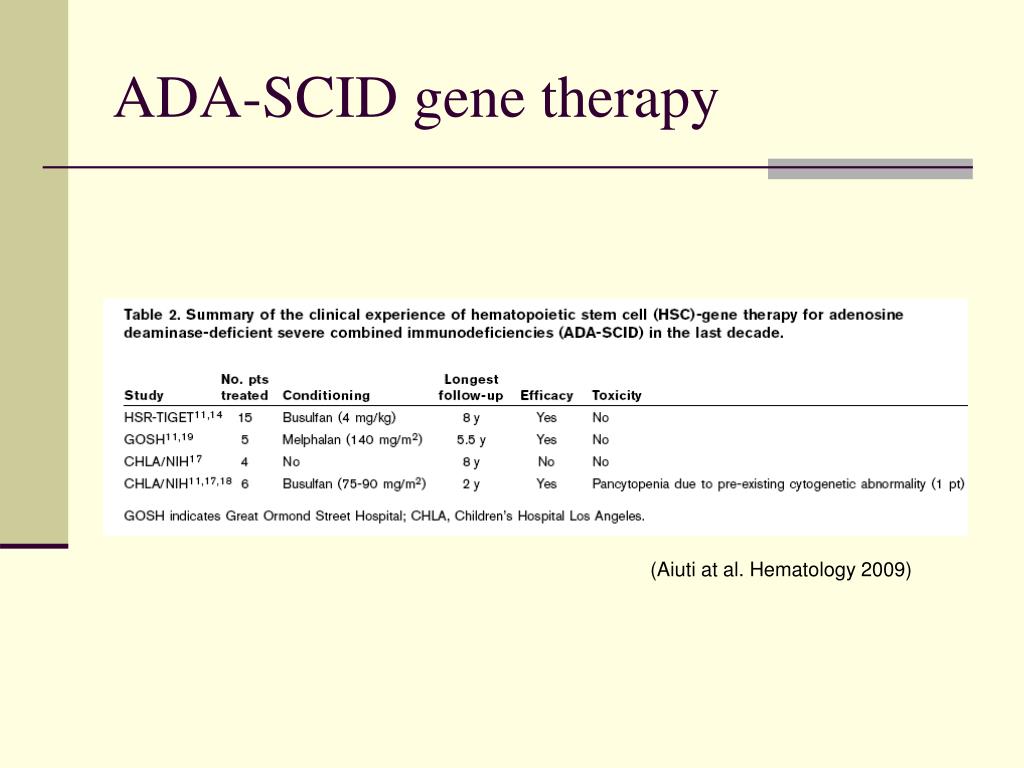
Without treatment, patients rarely survive toddlerhood. At the time, in 1990, she was a 4-year-old living with severe combined immunodeficiency (SCID), which is caused by insufficient levels of the enzyme adenosine deaminase (ADA). Despite slow progress and unexpected setbacks, the future of gene therapy remains bright.Īshanthi de Silva was the first human to be treated successfully with gene therapy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
